The selection of patient-reported outcomes (PROs) and clinician-reported outcomes (ClinROs) is crucial for the results of clinical trials investigating atopic dermatitis (AD). The top 3 clinical outcome assessments (COAs) are:
1. Eczema Severity Index (EASI)
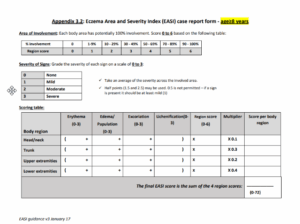
The EASI quantifies the severity of a subject’s AD based on both lesion severity and the percentage of body surface area (BSA) affected. The EASI, when conducted by a trained investigator, typically takes around 6 minutes to complete. This instrument is a composite score ranging from 0 to 72. Four anatomical sites (or body regions)—head and neck, trunk, upper extremities, and lower extremities—are examined for erythema, edema/papulation, excoriation, and lichenification. The severity of these signs is evaluated on a 4-point scale (from 0 to 3), allowing for half-steps (e.g.: 1.5 and 2.5). The area affected by AD within each anatomical site is estimated as a percentage (from 0% to 100%) of the total area and assigned a numerical value (from 0 to 6) based on the extent of involvement. A score per region is then calculated by multiplying the sum of the regional intensity score by the regional area score and the region-specific multiplier. Of note, the multiplier allows adjustment of the proportion of each body region to the whole body (and this is different for subjects under and above 8 years old). The final EASI score is the sum of the 4 body regions.
HOME— Harmonizing Outcome Measures for Eczema
2. Body Surface Area (BSA)
The evaluation of the overall BSA affected by AD ranges from 0% to 100%. Different methods exist to evaluate the BSA affected by AD, including:
- The palmar surface of one hand (using the patient’s hand), including the fingers, represents 1% of the total BSA. The investigator then calculates the total BSA involvement by counting the number of handprints with AD across the patient’s body.
- The Rule of Nines method for estimating the extent of AD lesions allocates percentages to various body parts. The whole head and neck account for 9% (split equally between the front and back at 4.5% each). The total trunk is estimated to be 36%, which can be further divided into 18% for the front and 18% for the back, upper and lower limbs account for 9% each and 18% each respectively. The remaining 1% is attributed to genitals.
3. Validated Investigator Global Assessment for AD (vIGA-AD)
The vIGA-AD™ is a static evaluation tool for morphological assessment of overall AD severity. The ordinal 5-point scale, spanning severity grades from 0 (clear) to 4 (severe), employs clinically relevant descriptions to alleviate variability among observers.
Special mention SCORing AD (SCORAD)
The SCORAD is a clinical tool for assessing the severity (i.e., extent, intensity) of AD as objectively as possible, and it is also the only COA that includes both a PRO and a ClinRO component. The AD severity can be assessed through these 6 signs: erythema, edema/papulation, oozing/crusts, excoriation, lichenification, and dryness, each graded on a 4-point scale (0 [absence] to 3 [severe], and half-steps are not allowed).
The individual intensity ratings are then added (ranging between 0 and 18) and multiplied by 3.5, giving a maximum score ranging between 0 and 63. The overall BSA affected by AD is evaluated on a scale from 0% to 100%, where the palmar surface of one hand is considered 1% of the total BSA. The overall BSA is divided by 5, giving a maximum score of 20. Patients are also asked to assess loss of sleep and pruritus each, using a 10 cm visual analog scale (0–10) based on the average of the last 3 nights, the combined score can range between 0 and 20. The cumulative score of the above measures represents the SCORAD, If the subjective scores of pruritus and loss of sleep are excluded, the SCORAD becomes objective SCORAD (score ranges 0–83).
It should be noted that there are ongoing efforts aiming to standardize and minimize the inaccuracies of BSA evaluations. Using a single EASI-derived BSA rather than taking up to 3 different BSA measurements per patient visit as usual practice could reduce the likelihood of error.
Table 1. Characteristics of Top 3 COA used in AD Trials
| Instrument | Global Assessment | ClinRO | PRO* | Validated in Adults | Validated in Children/Adolescent | License/
Permission Requirement |
Reference |
| EASI | ✔ | ✔ | ✔ | ✔ | Yes | Hanifin 2001 | |
| BSA | ✔ | ✔ | ✔ | ✔ | No | Schmitt 2014 Moore 2022 | |
| vIGA-AD | ✔ | ✔ | ✔ | Yes | Simpson 2020
Copyright © 2017 Eli Lilly and Company |
*It should be noted that PROs are also used in AD trials. This encompasses widely used instruments such as the Peak Pruritus Numeric Rating Scale (PP-NRS), Patient-Oriented Eczema Measure (POEM), and Dermatology Life Quality Index (DLQI) to gauge AD from the patient’s perspective.
Conclusion
In conclusion, the top 3 COAs currently used in AD trials are the EASI, BSA, and the vIGA-AD. These tools provide a comprehensive evaluation of AD severity, taking into account various factors such as lesion severity, the percentage of BSA affected, and overall morphological assessment. Special attention should be given to SCORAD, as it is currently the only COA that includes both a PRO and a ClinRO component.
Additionally, this article underscores the value of PROs in understanding the patient’s perspective, with instruments like PP-NRS, POEM, and DLQI being commonly used in AD studies. These assessments collectively contribute to a holistic understanding of AD and its impacts, thereby playing a pivotal role in the success of clinical trials.
About the author
A seasoned scientist and medical writing expert, Teresa is dedicated to fostering scientific engagement and enhancing the strategic value of medical affairs and innovation in clinical trials. She seamlessly navigates her roles with a decade-long background in preclinical research across various therapeutic areas.